On Wednesday, Kendric Cromer, a 12-year-old boy from a suburb of Washington, became the first person in the world with sickle cell disease to begin a commercially approved gene therapy that may cure the condition.
For the estimated 20,000 people with sickle cell in the United States who qualify for the treatment, the start of Kendric’s monthslong medical journey may offer hope. But it also signals the difficulties patients face as they seek a pair of new sickle cell treatments.
For a lucky few, like Kendric, the treatment could make possible lives they have longed for. A solemn and shy adolescent, he had learned that ordinary activities — riding a bike, going outside on a cold day, playing soccer — could bring on episodes of searing pain.
“Sickle cell always steals my dreams and interrupts all the things I want to do,” he said. Now he feels as if he has a chance for a normal life.
Near the end of last year, the Food and Drug Administration gave two companies authorization to sell gene therapy to people with sickle cell disease — a genetic disorder of red blood cells that causes debilitating pain and other medical problems. An estimated 100,000 people in the United States have sickle cell, most of them Black. People are born with the disease when they inherit the mutated gene for the condition from each parent.
The treatment helped patients in clinical trials, but Kendric is the first commercial patient for Bluebird Bio, a Somerville, Mass., company. Another other company, Vertex Therapeutics of Boston, declined to say if it had started treatment for any patients with its approved CRISPR gene-editing-based remedy.
Kendric — whose family’s health insurance agreed to cover the procedure — began his treatment at Children’s National Hospital in Washington. Wednesday’s treatment was only the first step. Doctors removed his bone marrow stem cells, which Bluebird will then genetically modify in a specialized lab for his treatment.
That will take months. But before it begins, Bluebird needs hundreds of millions of stem cells from Kendric, and if the first collection — taking six to eight hours — is not sufficient, the company will try once or twice more.
If it still doesn’t have enough, Kendric will have to spend another month in preparation for another stem cell extraction.
The whole process is so involved and time consuming that Bluebird estimates it can treat the cells of only 85 to 105 patients each year — and that includes not just sickle cell patients, but also patients with a much rarer disease — beta thalassemia — who can receive a similar gene therapy.
Medical centers also have the capacity to handle only a limited number of gene therapy patients. Each person needs expert and intensive care. After a patient’s stem cells have been treated, the patient has to stay in the hospital for a month. For most of that time, patients are severely ill from powerful chemotherapy.
Children’s National can accept only about 10 gene therapy patients a year.
“This is a big effort,” said Dr. David Jacobsohn, chief of the medical center’s division of blood and marrow transplantation.
Top of the Waiting List
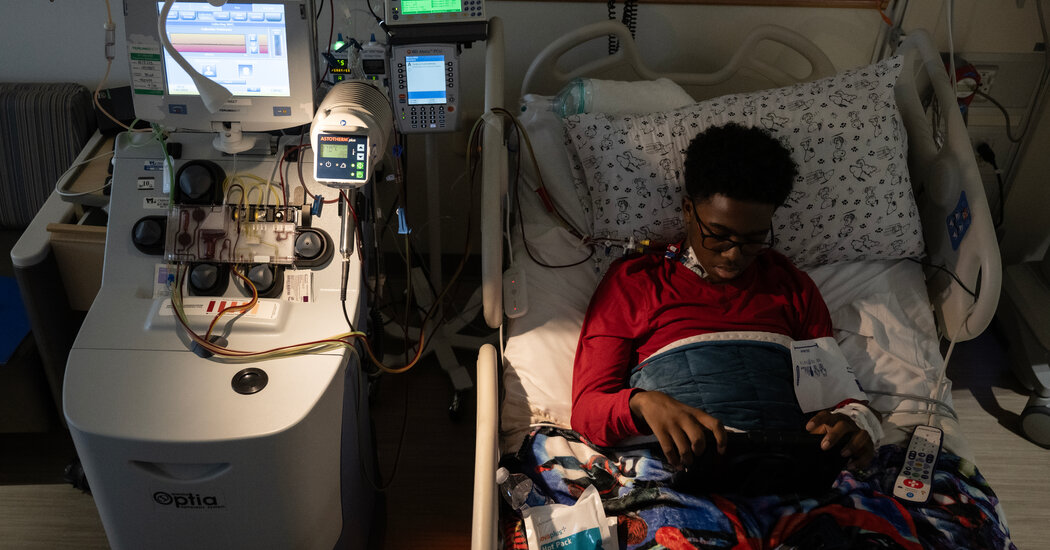
Last week, Kendric came prepared for the stem cell collection — he has spent many weeks in this hospital being treated for pain so severe that on his last visit, even morphine and oxycodone could not control it. He brought his special pillow with a Snoopy pillowcase that his grandmother gave him and his special Spider-Man blanket. And he had a goal.
“I want to be cured,” he said.
Bone marrow stem cells, the source of all the body’s red and white blood cells, are normally nestled in a person’s bone marrow. But Kendric’s doctors infused him with a drug, plerixafor, which pried them loose and let them float in his circulatory system.
To isolate the stem cells, staff members at the hospital inserted a catheter into a vein in Kendric’s chest and attached it to an apheresis machine, a boxlike device next to his hospital bed. It spins blood, separating it into layers — a plasma layer, a red cell layer and a stem cell layer.
Once enough stem cells have been gathered, they will be sent to Bluebird’s lab in Allentown, N.J., where technicians will add a healthy hemoglobin gene to correct the mutated ones that are causing his sickle cell disease.
They will send the modified cells back three months later. The goal is to give Kendric red blood cells that will not turn into fragile crescent shapes and get caught in his blood vessels and organs.
Although it takes just a couple of days to add a new gene to stem cells, it takes weeks to complete tests for purity, potency and safety. Technicians have to grow the cells in the lab before doing these tests.
Bluebird lists a price of $3.1 million for its gene therapy, called Lyfgenia. It’s one of the highest prices ever for a treatment.
Despite the astronomical price and the grueling process, medical centers have waiting lists of patients hoping for relief from a disease that can cause strokes, organ damage, bone damage, episodes of agonizing pain and shortened lives.
At Children’s National, Dr. Jacobsohn said at least 20 patients were eligible and interested. The choice of who would go first came down to who was sickest, and whose insurance came through.
Kendric qualified on both counts. But even though his insurance was quick to approve the treatment, the insurance payments are only part of what it will cost his family.
Chances and Hopes
Deborah Cromer, a realtor, and her husband, Keith, who works in law enforcement for the federal government, had no idea they might have a child with sickle cell.
They found out only when Deborah was pregnant with Kendric. Tests showed that their baby would have a one-in-four chance of inheriting the mutated gene from each parent and having sickle cell disease. They could terminate the pregnancy or take a chance.
They decided to take a chance.
The news that Kendric had sickle cell was devastating.
He had his first crisis when he was 3. Sickled blood cells had become trapped in his legs and feet. Their baby was inconsolable, in such pain that Deborah couldn’t even touch him.
She and Keith took him to Children’s National.
“Little did we know that that was our introduction to many many E.R. visits” Deborah said.
The pain crises became more and more severe. It seemed as though anything could set them off — 10 minutes of playing volleyball, a dip in a swimming pool. And when they occurred, Kendric sometimes needed five days to a week of treatment in the hospital to control his pain.
His parents always stayed with him. Deborah slept on a narrow bench in the hospital room. Keith slept in a chair.
“We’d never dream of leaving him.” Deborah said.
Eventually the disease began wreaking severe damage. Kendric developed avascular necrosis in his hips — bone death that occurs when bone is deprived of blood. The condition spread to his back and shoulders. He began taking a large daily dose of gabapentin, a medicine for nerve pain.
His pain never let up. One day he said to Deborah, “Mommy, I’m in pain every single day.”
Kendric wants to be like other kids, but fear of pain crises has held him back. He became increasingly sedentary, spending his days on his iPad, watching anime or building elaborate Lego structures.
Despite his many absences, Kendric kept up in school, maintaining an A average.
Deborah and Keith began to hope for gene therapy. But when they found out what it would cost, they lost some of their hope.
But their insurer approved the treatment in a few weeks, they said.
Now it has begun.
“We always prayed this day would come,” Deborah said. But, she added, “We’re nervous reading through the consents and what he will have to go through.”
Kendric, though, is looking forward to the future. He wants to be a geneticist.
And, he said, “I want to play basketball.”